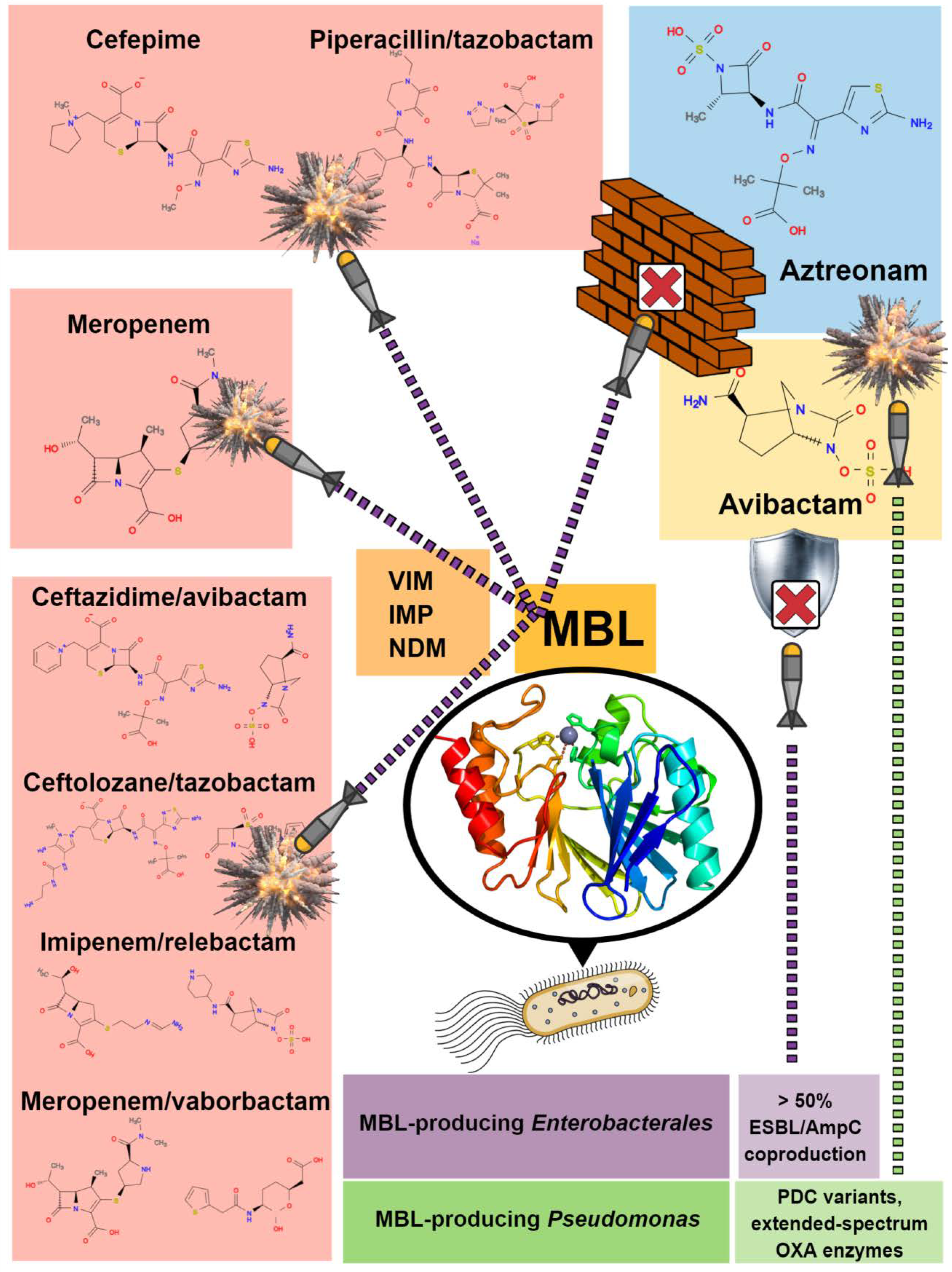
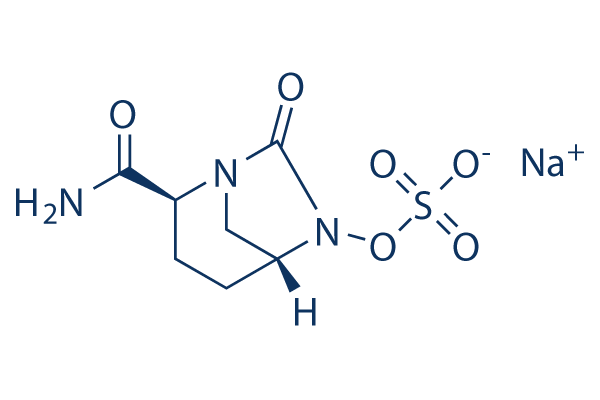
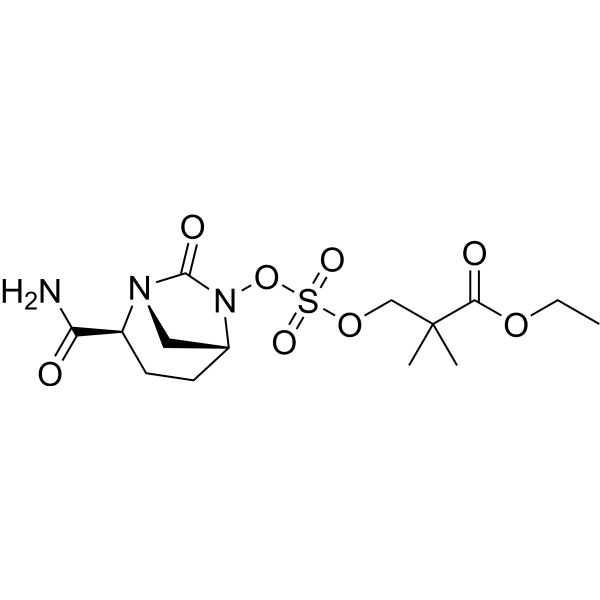
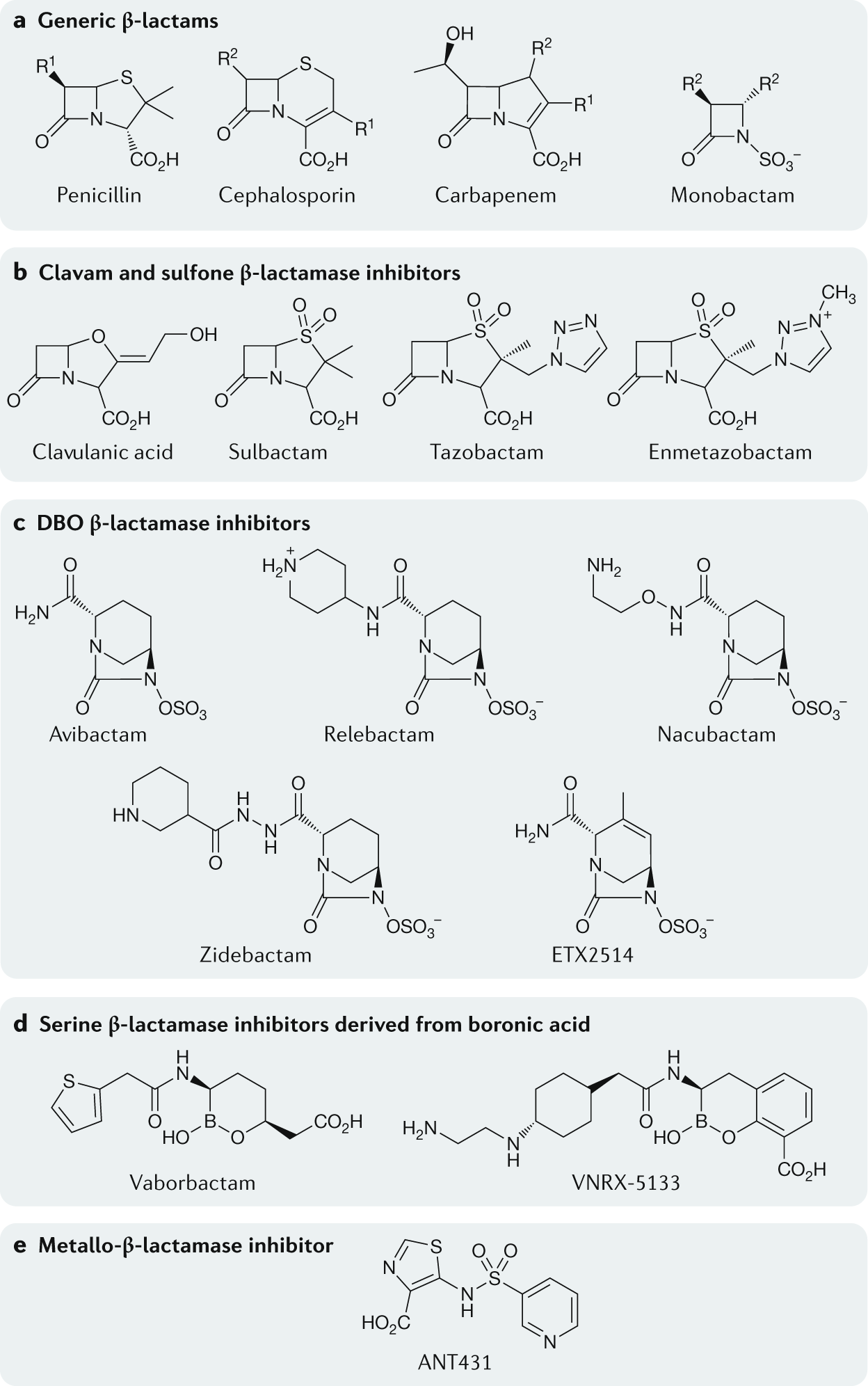
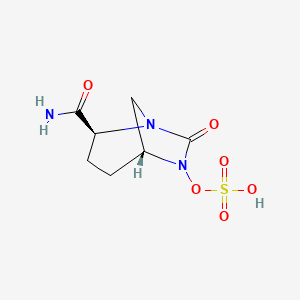
Molecular mechanisms underlying bacterial resistance to ceftazidime/ avibactam - Xiong - 2022 - WIREs Mechanisms of Disease - Wiley Online Library

Antibiotics - Beta lactam & beta lactamase inhibitor combinations: Nursing Pharmacology - Osmosis Video Library

Unexpected Challenges in Treating Multidrug-Resistant Gram-Negative Bacteria: Resistance to Ceftazidime-Avibactam in Archived Isolates of Pseudomonas aeruginosa | Antimicrobial Agents and Chemotherapy

CARIOU Kevin on Twitter: "Very happy to share that our first paper of 2020 is out! A review in @OrgBiomolChem on the synthesis of avibactam and other diazabicyclooctane β-lactamase inhibitors. Congrats to

A theoretical approach for the acylation/deacylation mechanisms of avibactam in the reversible inhibition of KPC-2 | SpringerLink

The road to avibactam: the first clinically useful non-β-lactam working somewhat like a β-lactam | Future Medicinal Chemistry
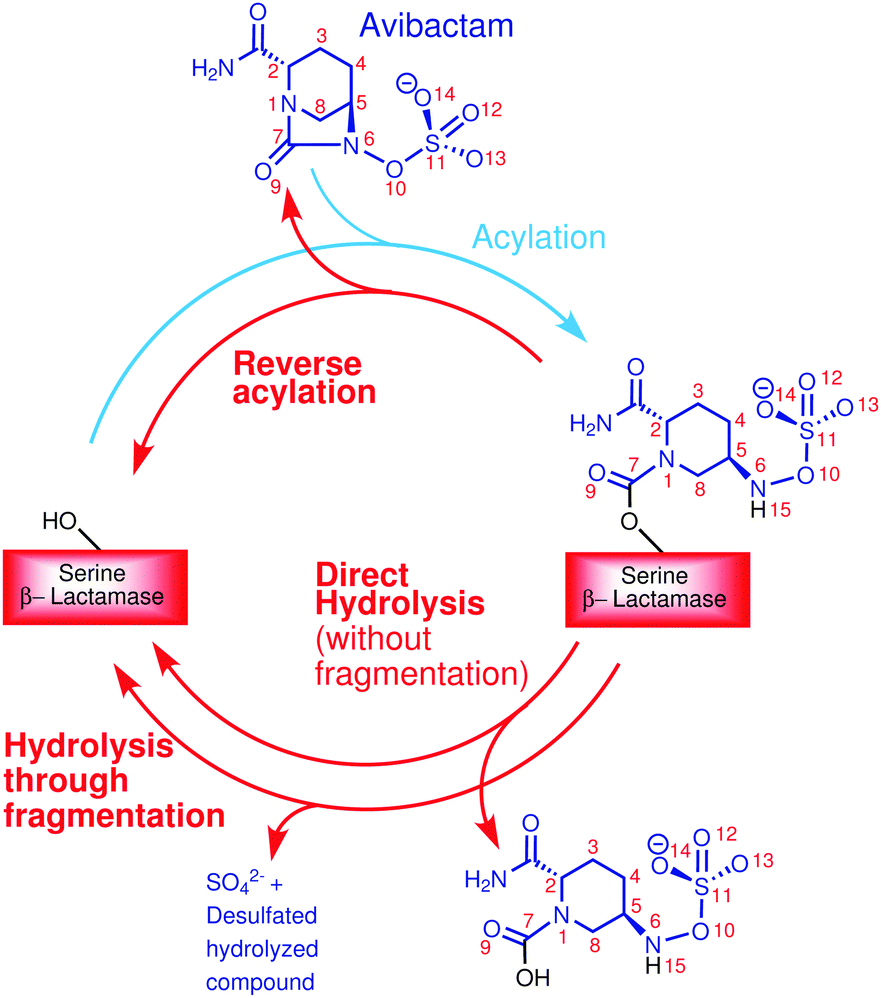
Molecular insights into avibactam mediated class C β-lactamase inhibition: competition between reverse acylation and hydrolysis through desulfation - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C8CP01670D